Wearable ECG QT Risk Checker
Typical raw QT ranges from 350ms to 450ms depending on heart rate.
Accuracy Note
Studies confirm devices like Apple Watch offer accuracy within a ±20ms margin of clinical 12-lead ECGs.
Corrected QT (Bazett Formula)
Standard formula used by most apps:
QTc = QT / √RR
Corrected QT (Fridericia Formula)
Alternative formula, better at high heart rates:
QTc = QT / (RR)1/3
Status Evaluation
Adjust the sliders above to calculate risk.
How to Interpret Results
| Risk Level | QTc Range | Clinical Context |
|---|---|---|
| Safe Zone | < 450ms | Normal range. Low risk of arrhythmia. |
| Borderline | 450ms - 480ms | Prolongation detected. Monitor closely, especially with new medications. |
| High Risk | > 480ms - 500ms+ | Significant prolongation. Potential risk for Torsades de Pointes (AI often flags >500ms). |
Key Takeaways
The Essentials of Wearable Cardiac Monitoring
- Wearable ECGs have gained FDA clearance for measuring QT intervals, a critical safety marker for heart rhythm disorders.
- Studies confirm devices like Apple Watch and KardiaMobile offer accuracy comparable to clinical 12-lead ECGs within a ±20ms margin.
- Artificial Intelligence models are now automating risk detection, reducing reliance on manual clinician review.
- Regulatory guidance issued during 2020 accelerated the adoption of remote monitoring for drug-induced arrhythmias.
The Urgent Need for Better Heart Rhythm Tracking
If you've ever worried about a medication affecting your heart, you aren't alone. Many common drugs, including certain antibiotics and antipsychotics, can alter your heart's electrical cycle. Specifically, they can lengthen the time between the Q wave and the T wave, known as the QT Interval. This interval represents the time for ventricular depolarization and repolarization. When this window gets too long, it increases the risk of a dangerous condition called torsades de pointes. Historically, catching this required hospital visits and expensive machines. Today, the landscape has shifted dramatically toward home monitoring.
The core problem remains consistent: how do we detect this subtle electrical change before it becomes a crisis? Traditional methods rely on static snapshots in a clinic. However, risks often happen when you're at home, sleeping, or under stress. That is where Wearable ECGs come into play. These consumer-grade devices have evolved from novelty trackers into medical tools capable of real-time risk detection. By wearing a sensor or using a handheld clip, users generate continuous data streams. This shift isn't just about convenience; it's about closing the gap between prescription and physiological impact.
How Consumer Devices Measure Professional Metrics
You might assume a wristwatch cannot replicate a hospital machine. Surprisingly, validation studies suggest otherwise. Several key devices have undergone rigorous testing to prove their reliability. The market leaders currently dominating the space include smartwatches like the Apple Watch and dedicated pocket ECGs like the KardiaMobile.
| Device Type | Lead Configuration | Accuracy Metric | Primary Use Case |
|---|---|---|---|
| Smartwatch (Single-Lead) | Lead I equivalent | Coefficient ~0.88-0.91 | Screening & Monitoring |
| Handheld Multi-Lead | Leads I, II, III, aVF, aVL, aVR | Within ±20 ms of Standard | Clinical Validation |
| Holter Monitor (Traditional) | 3+ Leads | Gold Standard Baseline | Long-term Diagnostic |
Let's break down what those numbers mean. In a study published by Spaccarotella in 2021, researchers compared smartwatch readings against standard 12-lead ECGs. They found Spearman's correlation coefficients reaching 0.914 for the mean QT interval. That is a very high statistical agreement. Even better, the Bland-Altman analysis showed reliability across different correction formulas, including Bazett and Fridericia. These formulas adjust for heart rate, which matters because your QT changes when you run versus when you rest. If a device fails to account for this, false alarms become common.
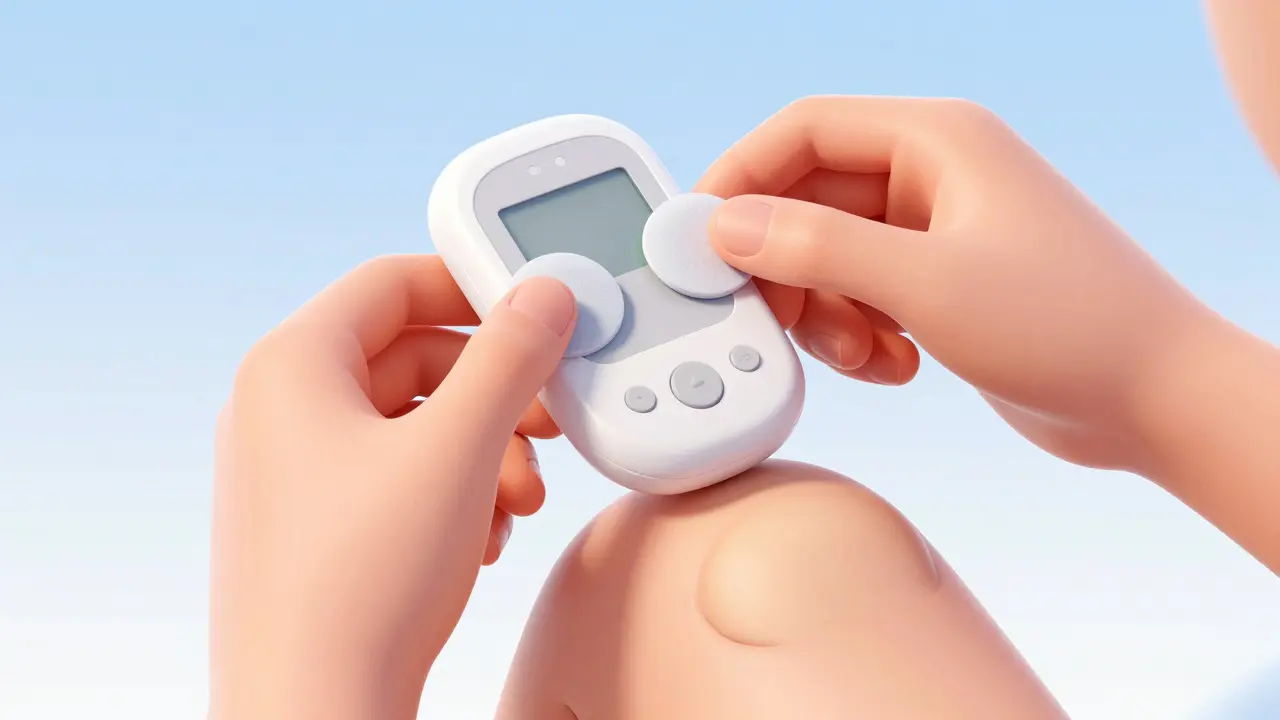
Another critical player is the KardiaMobile 6L. Unlike a watch worn on the wrist, this handheld unit uses three electrodes on each side. To get a reading, you touch the top with your thumbs and place the bottom on your knee or ankle. This creates a circuit that mimics a full chest setup. Pilot data from 2024 indicates this 6-lead device provides interval measurements comparable to a standard clinical ECG. It achieves this by capturing signals from multiple angles, reducing noise and improving the fidelity of the QRS complex detection.
Navigating the Regulatory Landscape
Technology moves fast, but regulations move slower. The path to approval involved significant hurdles. For years, FDA guidance was cautious about approving consumer gadgets for diagnostic metrics like the QT interval. The turning point arrived during the global health crisis of 2020. Hospitals were overwhelmed, and doctors needed ways to monitor patients receiving hydroxychloroquine and azithromycin from afar. These medications carry known risks for heart rhythm issues.
In April 2020, regulatory bodies issued specific permissions allowing mobile cardiac telemetry units for QT measurement. This wasn't just a suggestion; it opened a door for formal validation. As noted in documentation by Hoek et al. (2023), sixteen studies specifically examined the KardiaMobile 6L for QT measurements following this era. The regulatory stance acknowledged that while skin-to-electrode impedance can cause signal quality issues, the data remains generally consistent with other monitoring technologies. This acceptance paved the way for broader use in clinical trials.
Today, pharmaceutical sponsors increasingly use wearables for cardiac safety monitoring in Phase I-III trials. Why spend weeks sending patients back to a lab when they can upload data from home? It reduces patient burden and improves data completeness. However, compliance is still king. A device is only as good as the user's ability to hold it still and clean the contacts. Training participants is just as important as the hardware itself.
The Role of Artificial Intelligence in Detection
Hardware solves half the problem; software solves the rest. The biggest bottleneck in wearable monitoring is the sheer volume of data. One person generates thousands of beats per day. Who looks at them? Traditionally, a cardiologist would manually review recordings. This is slow and resource-intensive. Enter Artificial Intelligence.
Recent research by Alam et al. (2024) highlights a major leap forward. They developed a deep learning model using a Residual Neural Network. This system doesn't just look at heartbeats; it infers QT intervals from single-lead inputs automatically. The model processes ECG beats from Lead-I and Lead-II streams to predict QTc prolongation (defined as greater than 500ms). When tested on 686 patients with genetic heart disease, the results were promising. Half of these patients had long QT syndrome, a difficult subgroup to diagnose accurately.
Why does AI matter here? Because it enables automated QT monitoring out-of-hospital care. Imagine a scenario where a patient starts a new antibiotic. An algorithm flags a trending increase in their QT interval via an app notification before symptoms appear. That is the definition of preventative care. While manual review remains the gold standard for legal liability, AI handles the screening burden. It acts as a triage nurse, flagging only the concerning traces for human eyes. This hybrid approach ensures speed without sacrificing oversight.

Limitations and Practical Reality Checks
Despite the excitement, you must remain realistic about current capabilities. Not every heart issue is visible through these devices. A review in the Cleveland Clinic Journal of Medicine (2024) pointed out a specific weakness. While sensitivity for atrial fibrillation is excellent, the detection of pathologic Q waves dropped significantly. In some studies, sensitivity for these markers was only around 20.6% on consumer devices. This means if you are looking for signs of old heart attacks rather than current rhythm problems, wearables might miss them.
Signal quality is another factor. You can't just slap a device on sweaty skin after a workout and expect a perfect readout. Skin-to-electrode impedance affects signal integrity. Devices like the Apple Watch require you to press your finger on the digital crown to complete the circuit. If that contact is loose, the waveform wobbles. KardiaMobile requires limb contact, which helps but demands patience. High-quality data requires calm conditions and proper placement.
Furthermore, interpretation varies. There is currently no commercially available universal QT algorithm embedded in all consumer apps yet. Some manufacturers provide estimates, but they differ in how they calculate the T-wave end. This variability is why Dr. Sarah Handzel and others emphasize that while in-home options are growing, clinical validation for specific QT monitoring remains selective. Always consult a specialist who understands the limitations of the specific device you are using.
Frequently Asked Questions
Can a smartwatch replace a standard ECG in the doctor's office?
Not entirely. While studies show high correlation for specific metrics like the QT interval, smartwatches lack the multi-lead depth of a full 12-lead ECG. They are excellent for screening and longitudinal monitoring, but definitive diagnosis usually requires clinical equipment.
Which wearable device is best for QT monitoring?
Devices like the KardiaMobile 6L offer 6-lead recording, providing a better simulation of a clinical ECG compared to single-lead smartwatches. However, smartwatches like the Apple Watch Series 4 and later are FDA-cleared and offer convenient continuous tracking.
Does insurance cover wearable ECG monitors?
Coverage varies widely. Medical-grade devices prescribed for specific conditions are more likely to be covered than general wellness smartwatches. Check with your provider regarding telehealth coverage codes for remote monitoring devices.
How accurate are the readings compared to hospital machines?
For QT intervals, accuracy is generally within ±20 milliseconds of a standard ECG. Correlation coefficients in validation studies often exceed 0.88, indicating strong agreement for rhythm and interval timing.
Can these devices detect life-threatening arrhythmias?
Yes, many devices are cleared for detecting Atrial Fibrillation (AFib) and irregular rhythms. However, detection of ventricular arrhythmias like Torsades de Pointes relies on specific algorithmic updates that may not be present on all older models.


emma ruth rodriguez
March 31, 2026 AT 02:23The study references provided in the initial documentation are quite comprehensive, however, there is a need for further longitudinal analysis regarding signal drift over extended periods of wear time; furthermore, we must scrutinize the methodology behind the Bazett correction formula application in these consumer contexts because heart rate variability introduces significant noise.
Jonathan Sanders
April 1, 2026 AT 18:11Oh wonderful another piece of hardware to charge every morning instead of getting sleep
Kendell Callaway Mooney
April 1, 2026 AT 23:27I understand the frustration with charging but the real benefit lies in catching rhythm issues early which saves trips to the ER and prevents bad outcomes down the road for people at risk
Charles Rogers
April 3, 2026 AT 01:37I have been following the progress on cardiac telemetry for quite some time now. It is refreshing to see that validation data supports what we suspected was possible. However, the reliance on consumer electronics for medical diagnostics remains a precarious balance between accessibility and safety. The margin of error being twenty milliseconds sounds acceptable until you realize how fast ventricular repolarization actually occurs in pathological states. We must consider the implications when a patient relies solely on wrist-based sensors for life-critical intervention decisions. Insurance reimbursement codes have not caught up with the technology development at all. Many patients will find themselves paying out of pocket for devices that claim clinical validity. The regulatory guidance from two thousand twenty was indeed a pivotal moment for the industry sector. Yet compliance training for end-users is often neglected in favor of marketing hype regarding ease of use. Signal quality degradation due to sweat or movement artifacts is a genuine concern for continuous monitoring protocols. Algorithm updates require rigorous peer review before deployment to prevent false negatives in arrhythmia detection. It is crucial that we distinguish between wellness tracking and actual diagnostic capability in every conversation. Manufacturers need to be transparent about the specific lead configurations used in their testing protocols. The difference between a single lead and a six-lead setup changes the clinical utility significantly. We cannot allow the term FDA cleared to become a blanket endorsement of absolute medical equivalence. Patients deserve accurate information regarding the limitations of these wearable technologies.
Beccy Smart
April 3, 2026 AT 04:13Too much reading for me 😴 maybe just buy the watch 🙄
Christopher Curcio
April 4, 2026 AT 08:53From a technical standpoint the QRS complex fidelity matters immensely when interpreting the ST segment elevation potential in ischemic events versus simple QT prolongation which these single-lead models struggle to capture adequately without limb placement adjustment
RONALD FOWLER
April 4, 2026 AT 21:35This shift towards home monitoring is exactly what we have been waiting for.
Angel Ahumada
April 5, 2026 AT 23:33most people dont understand the physics behind electrode impedance and skin contact variability which leads to garbage data interpretation by the proprietary algorithms embedded in the software
Jonathan Alexander
April 6, 2026 AT 11:56The dramatic implications for personal health ownership are staggering and frankly terrifying if the system fails during a critical arrhythmic event where seconds determine survival outcomes
dPhanen DhrubRaaj
April 7, 2026 AT 03:36cultural acceptance of health monitoring varies greatly depending on trust in local healthcare infrastructure and digital privacy laws
Vikash Ranjan
April 8, 2026 AT 22:20i disagree with the premise that trust is the main barrier because most consumers prioritize convenience over security regardless of the risks involved
Rick Jackson
April 9, 2026 AT 21:40the philosophical question remains whether continuous surveillance enhances human agency or merely creates dependency on external validation systems for biological function